The Cellular Microenvironment Lab
RESEARCH
Tumor stiffness is associated with poor cancer prognosis and metastasis, and extracellular matrix (ECM) remodeling in the activated stroma is important in regulating invasion and metastasis. The ECM changes rigidity, inducing cancer cells to become more invasive and more likely to metastasize. Tumor cells are subjected to mechanical stress from the ECM, and they can sense and respond to mechanical forces by transducing them into a biochemical signals, called mechanotransduction. In addition, the abnormal ECM in tumors can affect cancer progression by activating stromal cells, e.g. fibroblasts that accelerate tumor growth and treatment resistance. These activated stromal cells further stretch the ECM, causing a snowball effect.
The tumor microenvironment (TME) has specifically large impact on the expressed surface receptors, activated or silenced signaling pathways and will influence the therapeutic response. E.g. WNT signaling is essential for the profibrotic and tumorigenic behavior, and in fibroblasts, the ECM stiffness promote the production of different profibrotic mediators and ECM proteins. More discoveries on WNT functions related to the ECM, as well as downstream targets, are warranted. The influence of the stromal cell types and the stiffness of the ECM on the WNT activation pattern in the tumors, are therefore of greatest interest. This pathway is also an attractive target for anti-cancer therapies, especially in advanced primary tumors and metastatic cancers.
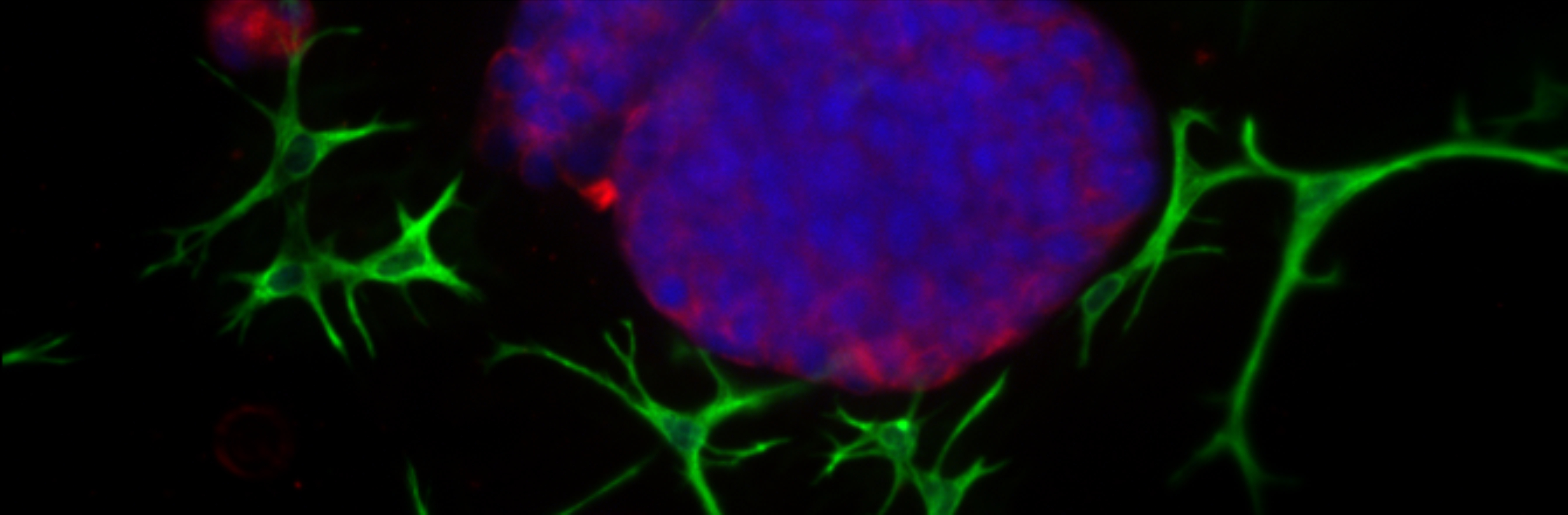
There is also lack of complex in vitro model systems that can recapitulate the complexity of ECM, tumor-stroma interactions and invasion, but be suitable for drug sensitivity testing. Thus, organotypic 3D co-culture models of epithelial tumor cells with e.g. fibroblasts in biologically relevant ECM have been established. They recapitulate the complex architecture observed in solid epithelial cancers, where “tumor islands” are surrounded and supported by connective tissue-like structures formed by the stromal cells. Different tumor/stroma co-culture settings in 3D can be probed with the various ECM preparations available. Tumor cells can be combined with relevant cell types typically found in the TME, and results can be brought into a wider context of cell-cell-interactions that play a role in solid cancers.
Patient-derived ex vivo models are important for determining the role of treatment-specific context. Protocols for the growth of primary tumor cells from clinical biopsies in organotypic 3D cultures have been established in the laboratory. Using confocal imaging and image analysis solutions, the effect of specific drug treatments on epithelial differentiation, cell-cell and cell-matrix contacts, polarization, EMT or invasion, can be quantified. The phenotypic assays has the future potential to help selecting correct treatment and tailoring combinations to that patient’s specific needs.
RESEARCH ENVIRONMENT
The research is carried out at Cell Biology, at Åbo Akademi University but it is also closely affiliated with the following research environments:
• FICAN West
• Turku Bioscience Screening Unit
• Turku Bioimaging
• Turku Cancer Research Society
COLLABORATORS
Important collaborators in ongoing research projects:
• Prof. Robert Kypta, CIC Biogune
• Prof. Juha Rantala, Sheffield University
• Prof. Cecilia Sahlgren, Åbo Akademi University
• Prof. Diana Toivola, Åbo Akademi University
• Prof. Jessica Rosenholm, Åbo Akademi University
• Prof. Pirkko Härkönen, University of Turku
• Prof. Matthias Nees, University of Lublin
PUBLICATIONS
Selected important publications:
Nassa G, Salvati A, Tarallo R, Gigantino V, Alexandrova E, Memoli D, Sellitto A, Rizzo F, Malanga D, Mirante T, Morelli E, Nees M, Åkerfelt M, Kangaspeska S, Nyman TA, Milanesi L, Giurato G, Weisz A. 2019. Inhibition of histone methyltransferase DOT1L silences ERα gene and blocks proliferation of antiestrogen-resistant breast cancer cells. Sci. Adv. 5:eaav5590.
Desai D, Åkerfelt M, Prabhakar N, Toriseva M, Näreoja T, Zhang J, Nees M, Rosenholm JM. 2018. Factors affecting intracellular delivery and release of hydrophilic vs hydrophobic cargo from mesoporous silica nanoparticles on 2D cellular and 3D tumor organoid level. Pharmaceutics 10: E237.
Björk JK, Ahonen I, Mirtti T, Erickson A, Rannikko A, Bützow A, Nordling S, Lundin J, Lundin M, Sistonen L, Nees M*, Åkerfelt M*. 2018. Increased HSF1 expression predicts shorter disease-specific survival of prostate cancer patients following radical prostatectomy. Oncotarget 9: 31200-31213.
Murillo-Garzón V, Gorroño-Etxebarria I, Åkerfelt M, Puustinen MC, Sistonen L, Nees M, Carton J, Waxman J, Kypta RM. 2018. Frizzled-8 integrates Wnt-11 and transforming growth factor-β signaling in prostate cancer. Nat. Commun. 9: 1747.
Ahonen I*, Åkerfelt M*, Toriseva M, Oswald E, Schüler J, Nees M. 2017. A high-content image analysis approach for quantitative measurements of chemosensitivity in patient-derived tumor microtissues. Sci. Rep. 7:6600.
Björk JK*, Åkerfelt M*, Joutsen J, Puustinen MC, Cheng F, Sistonen L# and Nees, M#. 2016. Heat-shock factor 2 is a suppressor of prostate cancer invasion. Oncogene 35: 1770-1784. *#
Robinson S, Guyon L, Nevalainen J, Toriseva M, Åkerfelt M* and Nees M*. 2015. Segmentation of image data from complex organotypic 3D models of cancer tissues using Markov random fields. Plos One 10: e0143798.
Åkerfelt M, Bayramoglu N, Robinson S, Toriseva M, Schukov H-P, Virtanen J, Härmä V, Kaakinen M, Kannala J, Eklund L, Heikkilä J, and Nees M. 2015. Automated tracking of tumor-stroma morphology in microtissues identifies targets within the tumor microenvironment for therapeutic intervention. Oncotarget 6: 30035-530056.
Härmä V, Schukov H-P, Happonen A, Ahonen I, Virtanen J, Siitari H, Åkerfelt M, Lötjönen J and Nees M. 2014. Quantification of dynamic morphological drug responses in 3D organotypic cell cultures by automated image analysis. Plos One 9: e96426.
CONTACT
If you are interested in a research collaboration or joining the laboratory, please contact:

Malin Åkerfelt, PhD
Docent in Translational Cancer Biology
University Lecturer in Cell Biology
Cell Biology
Faculty of Science and Engineering
Åbo Akademi University
Address: Biocity 2nd floor, Tykistökatu 6A, 20520 Turku, Finland
E-mail: malin.akerfelt@abo.fi
Phone: +358 50 3229868

