Inflammatory Signalling Lab

Research focus
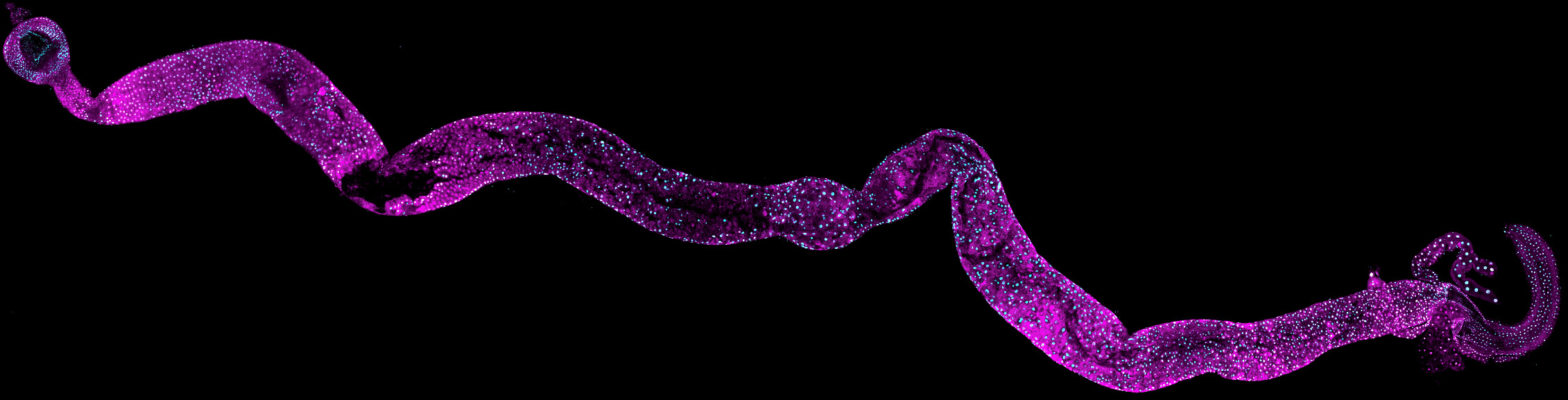
Chronic inflammatory intestinal diseases, which apprehensively are a major risk factor for colorectal cancer, are increasing in Europe and worldwide. Hence, we need mechanistic insights for molecular control and prevention of harmful inflammation. Inflammation is induced in the intestinal cells when they encounter pathogens and/or when they are exposed danger signals released from surrounding cells.
To protect themselves, the cells change their gene expression to produce factors promoting survival and proliferation such as cytokines, growth factors, antimicrobial peptides, and anti-apoptotic proteins. While this innate inflammatory response is required for cells and the organism to recover from infection, excess inflammatory signals may lead to loss of homeostasis, promoting chronic inflammation and cancer progression.
In the Inflammatory Signalling Group, we use mammalian and non-mammalian model systems to identify changes in and consequences of ubiquitin-modifications in the evolutionary conserved intestinal epithelial cells during local inflammation and cell stress. We aim to generate fundamental knowledge in how ubiquitin modifications are regulated and recognized, and how this signal is converted into inflammation-inducing activation of gene expression. Our goal is to control stress-induced and inflammation-associated survival and proliferation by regulating transcriptional responses to maintain cell integrity and tissue homeostasis.
Sammanfattning av forskningen på svenska
Sjukdomar som involverar kronisk inflammation, såsom inflammatorisk tarmsjukdom ökar globalt, särskilt bland unga människor. Samtidigt är kroniska tarminflammationer en riskfaktor för tjocktarmscancer. På motsvarande sätt kan kronisk inflammation i luftvägarna förorsakad av astma eller lungsjukdomar förknippas med lungcancer. Mitt laboratorium undersöker hur en proteinmodifikation som kallas ubikvitinering påverkar inflammation i cellerna i tarmen och luftvägarna. Eftersom inflammationssignalering är mycket komplex i däggdjursceller, använder vi bananflugan Drosophila melanogaster som modellorganism i våra studier. Vår forskning kommer att förbättra vår förståelse av hur proteinmodifikationer påverkar inflammationssignalering i cellerna och denna kunskap öppnar möjligheter för upptäckt av nya sätt att detektera och nya läkemedel för att hämma kronisk inflammation.

