
News
Researchers at Åbo Akademi University Contribute to Breakthrough in Needle-Free Glucose Monitoring
Researchers at Åbo Akademi University are contributing to the development of a new, needle-free method for measuring blood sugar. Adjunct Professor Zhanna Boeva and Professor Johan Bobacka are co-founders of the Finnish company GlucoModicum, which has attracted international attention with its glucose meter SOFIO.
The new glucose meter is based on a technology called magnetohydrodynamics (MHD), which has been patented by GlucoModicum. The meter uses a gentle electromagnetic pulse to bring a small amount of tissue fluid to the surface of the skin and then analyzes the glucose concentration in the fluid.
GGlucoModicum describes SOFIO as “the world’s first glucose magnet”.
The MHD technology was originally developed at the University of Helsinki, where researchers demonstrated that the method can extract tissue fluid significantly more efficiently than previous needle-free alternatives. In SOFIO, MHD is combined with a sensitive biosensor that measures the glucose content of the fluid. The sensor technology builds on long-standing expertise in chemical sensor research at Åbo Akademi University.
“We have extensive experience in research on electrochemical sensors at Åbo Akademi University, which has significantly contributed to the development of the sensor technology used here to analyze glucose levels. When our sensor research was combined with MHD technology from the University of Helsinki, it became possible to develop an entirely new type of glucose meter, something that had not been possible before,” explains Johan Bobacka.
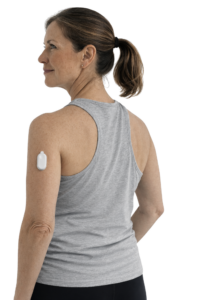
A particularly distinctive feature of SOFIO is its ease of use. The glucose meter has been developed especially for people with type 2 diabetes who do not need to wear a sensor continuously. Instead, users employ a small, reusable transmitter together with a soft sensor that is attached to the skin for a single day when they wish to check blood sugar levels.
Clinical studies involving more than 2,000 participants have shown that the technology meets accuracy requirements and that it can become a simpler and more flexible alternative to today’s glucose monitoring devices. The researchers believe that SOFIO will be a cost-effective option once the meter becomes commercially available.
The past year has brought several important milestones for GlucoModicum. In spring 2025, the company underwent a quality audit in accordance with the medical device standard ISO 13485, with zero nonconformities. The same year, the Finnish Medicines Agency (FIMEA) granted approval to proceed with a larger clinical study required for future approval and sales in Europe.

At the beginning of 2026, it was also confirmed that the company is collaborating with the UK-based firm Mpac Lambert to build a fully automated production line. The line will be able to manufacture up to 21 million sensors per year, which means the technology can reach users on a large scale if approved.
“The development of SOFIO has been both exciting and challenging. Creating a product in clinical diagnostics that also can be manufactured in millions of units requires enormous effort. At the same time, it has been an incredibly fascinating process, and not exactly something that we researchers deal with every day,” says Johan Bobacka.




